Traditional Caco-2 monolayer models fall short when predicting human-relevant intestinal toxicity, particularly for inflammation-driven responses like diarrhea. VivoSim’s NAMkind™ Intestine Model offers a breakthrough solution—combining physiological relevance with predictive power. Built on a 3D Transwell platform, the NAMkind™ Intestine Model incorporates both epithelial and stromal cells, enabling a more accurate recreation of native intestinal tissue and function. This advanced model allows for best-in-class diarrhea and inflammatory toxicity prediction, capturing key cytokine signaling that traditional models miss.
With integrated assays to measure epithelial barrier integrity (TEER) and differentiate between the viability of epithelial and stromal compartments, the NAMkind™ Intestine Model delivers a detailed and dynamic view of gut health under drug exposure, empowering researchers to make safer, more informed decisions earlier in development.
INTESTINAL TOXICITY TESTING FROM
OUR NAMkind INTESTINE MODEL
Fast. Cost-effective. Predictive.
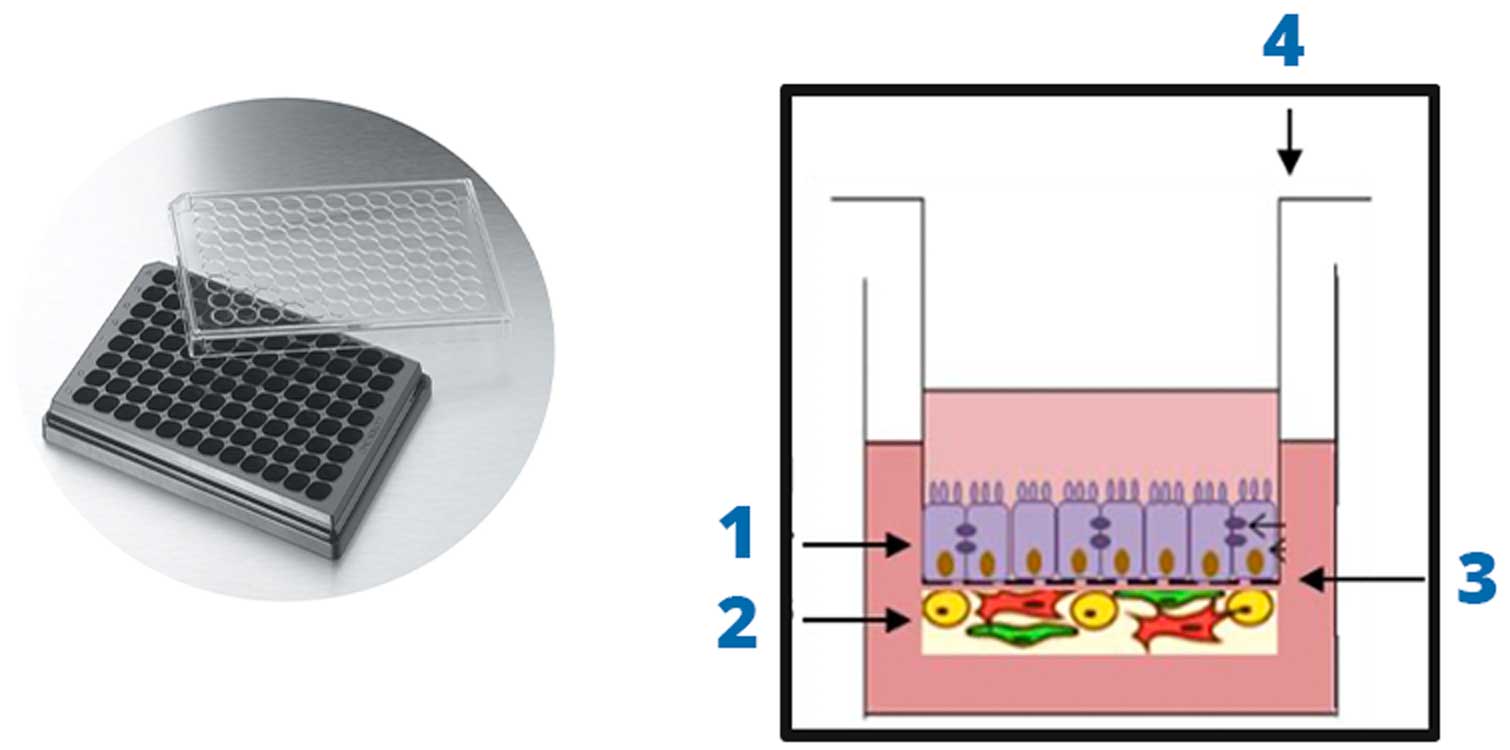
1. Epithelial Cell Layer
2. Stromal Cells
- Endothelial Cells
- Smooth Muscle Cells
- Transwell Insert
3. Permeable Membrane
4. Transwell Insert
Model Comparison
| NAMkind™ Intestine | Microfluidics (e.g., Intestine-On-Chip) |
Spheroid | Caco-2 Monolayer | |||
| Barrier Function | Cytotoxicity | |||||
| Barrier Renewal Capacity | ||||||
| Low Permeability | ||||||
| Barrier Integrity (TEER) | ||||||
| Biological Relevance | Multicellular | |||||
| ECM interaction | ||||||
| Practical Considerations | Throughput | |||||
| Ease of use | ||||||
| Cost Effective |
| NAMkind™ Intestine | ||||
|---|---|---|---|---|
| Barrier Function | Cytotoxicity | |||
| Barrier Function | Barrier Renewal Capacity | |||
| Barrier Function | Low Permeability | |||
| Barrier Function | Barrier Integrity (TEER) | |||
| NAMkind™ Intestine | ||||
|---|---|---|---|---|
| Biological Relevance | Multicellular | |||
| Barrier Function | ECM interaction | |||
| NAMkind™ Intestine | ||||
|---|---|---|---|---|
| Practical Considerations | Throughput | |||
| Practical Considerations | Ease of use | |||
| Practical Considerations | Cost Effective | |||
| Microfluidics (e.g., Intestine-On-Chip) |
||||
|---|---|---|---|---|
| Barrier Function | Cytotoxicity | |||
| Barrier Function | Barrier Renewal Capacity | |||
| Barrier Function | Low Permeability | |||
| Barrier Function | Barrier Integrity (TEER) | |||
| Microfluidics (e.g., Intestine-On-Chip) |
||||
|---|---|---|---|---|
| Biological Relevance | Multicellular | |||
| Barrier Function | ECM interaction | |||
| Microfluidics (e.g., Intestine-On-Chip) |
||||
|---|---|---|---|---|
| Practical Considerations | Throughput | |||
| Practical Considerations | Ease of use | |||
| Practical Considerations | Cost Effective | |||
| Spheroid | ||||
|---|---|---|---|---|
| Barrier Function | Cytotoxicity | |||
| Barrier Function | Barrier Renewal Capacity | |||
| Barrier Function | Low Permeability | |||
| Barrier Function | Barrier Integrity (TEER) | |||
| Spheroid | ||||
|---|---|---|---|---|
| Biological Relevance | Multicellular | |||
| Barrier Function | ECM interaction | |||
| Spheroid | ||||
|---|---|---|---|---|
| Practical Considerations | Throughput | |||
| Practical Considerations | Ease of use | |||
| Practical Considerations | Cost Effective | |||
| Caco-2 Monolayer | ||||
|---|---|---|---|---|
| Barrier Function | Cytotoxicity | |||
| Barrier Function | Barrier Renewal Capacity | |||
| Barrier Function | Low Permeability | |||
| Barrier Function | Barrier Integrity (TEER) | |||
| Caco-2 Monolayer | ||||
|---|---|---|---|---|
| Biological Relevance | Multicellular | |||
| Barrier Function | ECM interaction | |||
| Caco-2 Monolayer | ||||
|---|---|---|---|---|
| Practical Considerations | Throughput | |||
| Practical Considerations | Ease of use | |||
| Practical Considerations | Cost Effective | |||
Request Intestine Model Case Study and Data Report
Please fill out form to download intestine model case study
Publications
October, 2025
Development of a Multicellular 3D Intestinal Model Using Human Primary Cells to Identify Novel IBD Therapies
Gervacio S, Dudum R, Aidnick H, et al. Digestive Disease Week 2025, Abstract No. Tu2035
Read more »October, 2025
Identification of JAK Inhibitors as Potential Therapeutics for Inflammatory Bowel Disease Using Human Primary Cell 3D Models of Crohn’s Disease and Ulcerative Colitis
Dudum R, Payton O, Toohey C, et al. Digestive Disease Week 2025, Abstract No. Tu2040
Read more »FDA Alignment
Aligned with FDA’s New Approach Methodology (NAM) Framework
Our GI toxicity services are fully aligned with the FDA’s initiative to advance non-animal testing methods. NAMkind™ models are benchmarked against FDA and NIH reference compounds and adhere to performance criteria outlined in the Tox21 and NCATS collaborative frameworks for translational toxicology.
Our goal: Provide human-relevant intestinal data that meets the rigor of FDA expectations — supporting safer and faster drug development decisions.
VivoSim partners with pharmaceutical, biotech, and AI-driven drug discovery teams to modernize preclinical safety testing. By merging human primary cell biology, AI-powered analytics, and FDA-aligned NAM frameworks, we enable a more predictive, ethical, and efficient path to development

